- Piston Position Programme
- Example1
- Example2
- Offset Gudgeon Pin / Crankshaft
- Measurement of Port Timing
Piston Position Programme
The Piston Position Programme enables the user to calculate the piston position relative to tdc given the crank angle. Alternatively the crank angle can be calculated given the piston position relative to tdc. Hence it is possible to accurately measure the port timings for an engine.
This means that it is easy to check the "tune" of an engine and make decisions on if there is scope for further modification of the engine.
Top
Example1
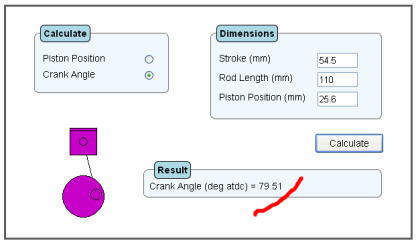
Suppose you have a two-stroke engine with a 54.5 mm stroke and a rod length of 110 mm. You wish to measure the exhaust port timing, so you remove the cylinder head and using a digital vernier you measure from the top of the cylinder down to the piston edge, with the exhaust port about to open. Now if this measurement is say 25.6mm you can use the above programme to calculate the crankangle at which the exhaust port opens.
You now have all the relevant information needed for the programme. Click on the crank angle button within the Calculate box. Now within the Dimensions box, enter 54.5 for the stroke, hit the tab key then enter 110 for the rod length, hit the tab key again and enter 25.6 for the piston position. Hit the calculate button. The answer is then calculated as 79.51 deg atdc.
Example2
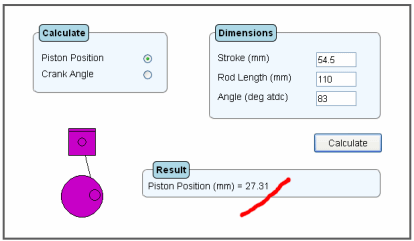
Now suppose you have the same two-stroke engine as the previous example. This time you require the distance from tdc that the exhaust port should open to give an opening angle of 83 deg atdc. Click on the piston position button within the Calculate box. Now within the Dimensions box, enter 54.5 for the stroke, hit the tab key then enter 110 for the rod length, hit the tab key again and enter 83 for the angle. Hit the calculate button. The answer is then calculated as 27.31 mm from tdc.
Offset Gudgeon Pin / Crankshaft
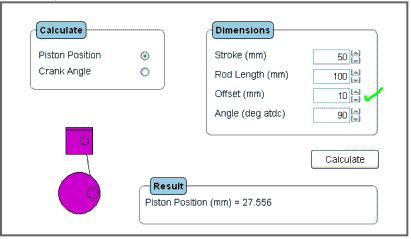
The "tuner" and "works tuner" versions of the piston position programme allow for the input of an offset gudgeon pin or offset crankshaft.
The figure below shows the output screen for an engine with a stroke of 50 mm, rod length 100 mm and an offset of 10 mm.
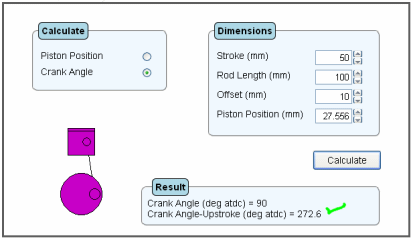
The figure below shows the output screen for the same engine parameters, but this time calculating the angle given a piston position of 27.556 mm. It is evident that the down-stroke angle is 90 deg atdc but the up-stroke angle is 272.6 deg atdc i.e the engine has asymmetric timing.
Consequently, if one engine has a 0 mm offset and a second engine has a 10 mm offset, and both engines open their exhaust ports at 90 deg atdc, the exhaust duration will be 180 degrees crank angle for the 0 mm offset engine and 182.6 degrees crank angle for the 10 mm offset engine.
Measurement of Port Timing
The photo shown below shows the measurement of the exhaust port opening for a late model Honda RS125. A feeler gauge is used to determine more precisely the opening point of the exhaust port. A measurement is taken from the top of the cylinder down to the edge of the piston. From this value must be subtracted the distance from the top of the cylinder down to the edge of the piston with the piston at tdc. Also, the thickness of the feeler gauge should be subtracted from this figure. Hence in this particular example the exhaust port opening height from tdc is 28.98 - 0.7 -0.2 = 28.08 mm.
